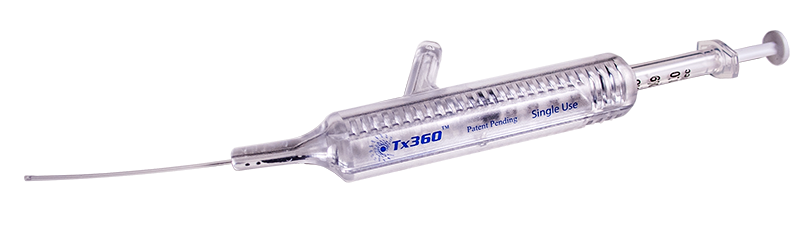
Tx360® Nasal Applicator for SPG Nerve Block
The Tx360® Nasal Applicator was developed by a biotech research and development company in the United States. This medical device is currently being manufactured in California, USA.
The Tx360 technology was cleared for use by the FDA in 2010, as a medical device for healthcare professional, to deliver small amounts of fluid to the nasal pathway, including the area of the inferior turbinate, superior turbinate, sphenopalatine foramen, etc. It is intended for use on adult patients with intact nasal mucosa. The device allows easy, safe and instant access to the inner most areas of the nasal pathway. It allows the healthcare professional to easily reach the Sphenopalatine foramen which is the targeted region, when performing a Sphenopalatine ganglion nerve block. To learn more about the TX360® Nasal Applicator Click Here.